First syncytial virus vaccine approved for pregnant women and people over 60
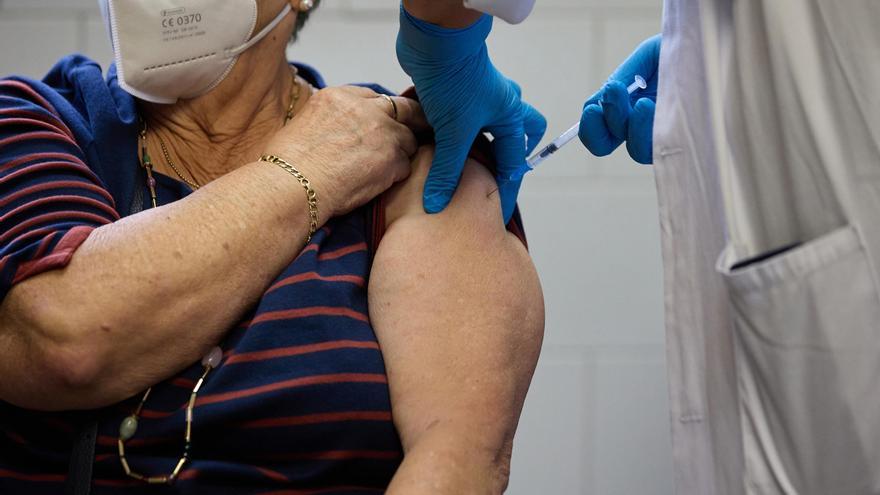
Pregnant women can now protect their babies under 6 months of age from bronchiolitis and other respiratory diseases by getting vaccinated between 24 and 36 weeks of pregnancy with the first adult serum that has just been approved by the Ministry of Health against Respiratory syncytial virus (VRS).
Bivalent vaccine Pfizer against prefusion protein F (RSVpreF), also indicated for those over 60 years oldbut for now the National Health System will fund it only for pregnant women, explained the company’s medical director, Jose Chavez, at the presentation of the drug.
Waiting for instructions that Public Health Commissionwho must now decide whether to include her in the lifelong vaccination schedule. Abrysvo, the commercial name of the vaccine, consists of a single dose that is injected into the shoulder muscle and is now available in pharmacies for €234.95. VAT included.
“Additional” to nirsevimab
Since October newborns immunized with nirsevimabA monoclonal antibody, not a vaccine, dramatically reduces hospitalizations due to RSV, which is the second leading cause of death in children under 12 months of age worldwide.
Supposedly in Spain Infections caused by this virus cause between 7,000 and 14,000 hospitalizations annually. that is, in 2% of cases, not forgetting that syncytial can lead to future consequences leading to symptoms similar to those of asthma in the first 5 years of life.
This vaccine, the first to arrive in Spain for adults and the only one indicated for pregnant women (the second vaccine developed by GSK is intended only for older people), is a new tool with which they will receive passive protection through the placenta of their children. mother.
According to Maria Maria Garcés, pediatrician at the Nazareth Medical Center (Valencia) and member of the Vaccine Advisory Committee of the Spanish Association of Vaccinologists (CAV-AEP), there should be “no interaction” between both drugs, which “may even be complementary.”
A vital shield for babies
“Just a few years ago Vaccination of a pregnant woman was a taboo topicbut thanks to scientific data, advances and industry, we have seen the importance of vaccination, first to protect ourselves from various diseases and then to the fetus,” said Inmaculada Cuesta, a nurse, midwife and secretary of the National Nurses Association. and vaccines (Anenvac).
When a woman is vaccinated, she produces antibodies, which she passes to the fetus through the placenta. “This is an altruistic act that a woman performs for the benefit of her child.“, which he gives a “vital shield” against RSV in the first six months of its life.
Since the child’s protection from RSV depends on the transfer of antibodies, Abrysvo should be prescribed between 24 and 36 weeks of pregnancy; What is important, the expert wanted to make clear, is that women of fertile age achieve pregnancy by following a full vaccination schedule, especially if they are women with any risk factor.
VRS is also something for adults
But HRV It’s not just for the little ones– explained Angel Gil, professor of preventive medicine and public health at the Juan Carlos University of Madrid – but it also has an impact on adults over 65 years of age, who are underdiagnosed, despite the fact that they have a higher mortality rate. nosocomial infection is associated with -8%- more cases of this virus than influenza.
90% of the population of this age has an underlying chronic disease, which becomes two at age 75 and at least three by age 80, so protecting them from any respiratory disease is “essential.”
Gil wanted to insist that the vaccine will not prevent infection, but it will prevent hospitalizations and deaths, as happens with other diseases such as Covid or the flu.
It may be caused by the flu
According to your prospectus, The spray can be placed parallel to vaccine seasonal flu. Like any drug, it has side effects, the most common in pregnant women are pain in the puncture area, headache, muscle pain and nausea.
In older adults, the most common symptoms were fatigue, headache, injection site pain, and myalgia.
With these two indications already approved, the pharmaceutical company got to work. two additional clinical studies to evaluate Abrysvo in children at increased risk of RSV disease aged 2 to 18 years.
The second study is evaluating adults ages 18 to 60 who are at increased risk for RSV due to underlying medical conditions such as asthma, diabetes, and chronic obstructive pulmonary disease (COPD), as well as adults ages 18 and older who are immunocompromised and Consequently, they are at greater risk of contracting RSV, concluded Jose Chavez, Pfizer’s medical director.
